|
11/7/2023 0 Comments Definition of sigma bond 
They can also be stable or unstable depending on whether electrons are in bonding or anti-bonding molecular orbitals. Sigma bonds and pi bonds are both based on certain molecular orbitals that are produced from the overlapping of specific atomic orbitals, such as s orbitals for sigma bonds and p orbitals for pi bonds. Similarities between sigma bonds and pi bonds P-P overlapping (Along the internuclear axis, one half-filled p orbital from each participating atom experiences head-on overlapping.).S-P overlapping (Along the internuclear axis, one half-filled s orbital overlaps with one half-filled p orbital, producing a covalent bond.).Before one s orbital may overlap with another, it must be half-filled. S-S overlapping (One ‘s’ orbital from each participating atom experiences head-on overlapping along the internuclear axis in this type of overlapping. 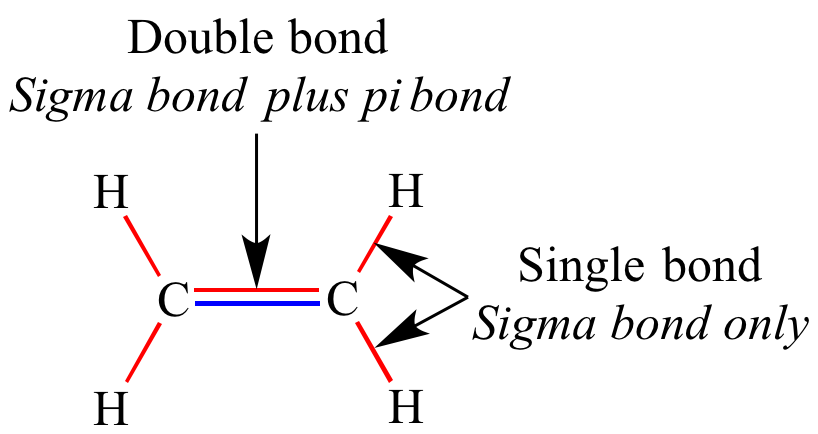
One sigma bond and one Pi bond combine to form a double bond. There are two double bonds in carbon dioxide. As a result, the sigma ( σ ) bond is more powerful than the Pi ( π) bond. The lateral overlap of the atomic orbitals that are aligned perpendicular to the internuclear axis forms a pi bond, thus the extent of orbital overlapping in a sideways manner is smaller. The pi bond is a covalent connection created by the lateral overlapping of half-filled atomic orbitals of atoms. The sigma bond is a covalent link produced by the head-on overlapping atomic orbitals. Differences between sigma bonds and pi bonds.Similarities between sigma bonds and pi bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
